NEWARK, Del., May 20, 2019 /PRNewswire/ — QPS, a global contract research organization (CRO) providing discovery, preclinical and clinical drug development services, today announced rapid growth in Europe fueled by a 170 percent increase of in vitro studies, with more than half of in vitro clients moving to perform follow-on in vivo studies with the company.
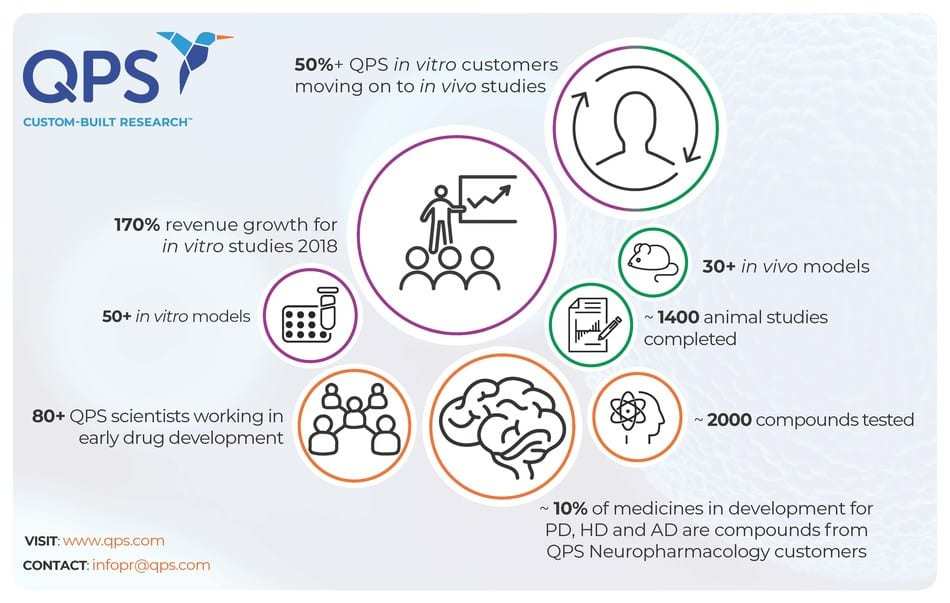
“We’re seeing a huge uptick in our in vitro research revenue because pharmaceutical and biotech companies seeking better outcomes are choosing to partner with CROs that have proven animal models and cell models that match,” said Birgit Hutter-Paier, Director, Neuropharmacology, QPS. “QPS is well-known for its transgenic mouse models, with more than 1,400 transgenic animal studies completed. That same expertise is required for in vitro studies. Therefore, wherever possible we translate our knowledge from in vivo models to our in vitro studies.”
More pharmaceutical and biotech companies are choosing QPS because the CRO designs its in vitro and follow-on in vivo studies using closely related models to, for instance, ensure the expression of targets are the same. For example, QPS has a mouse model expressing the same gene construct as its cell line.
“This strategy ensures our clients will test their compound in in vitro and in vivo models with the same promoter, expressing the same protein fragment, with the same mutations,” said Hutter-Paier. “The closer the in vitro model is to the in vivo model, the better the outcome.”
“When you consider the cost of in vivo studies, pharmaceutical and biotech companies can’t afford to test the wrong models in vitro. When they do, they run into problems when testing in vivo,” she continued. “Testing the wrong models is the biggest mistake we see companies make as they work to advance compounds from in vitro to in vivo. This happens most often when companies use one partner to conduct in vitro studies and then use a different partner to conduct in vivo studies.”
More and more companies are choosing a CRO partner that can take them from functional screening in vitro through in vivo and beyond, as evidenced by QPS’s rapid growth in this area.
“QPS scientists have leveraged the company’s successful transgenic mouse models to develop corresponding cell models. This strategy ensures our models work so we can move compounds to the next phase,” said Hutter-Paier.
QPS has tested thousands of CNS compounds at its preclinical site in Austria, with many of those compounds progressing through clinical development. In fact, CNS compounds from QPS Austria clients make up approximately 10 percent of all medicines currently in development for Parkinson’s disease, Huntington’s disease, as well as Alzheimer’s disease and other dementias.
For more insights on choosing a single CRO partner that can take companies from functional screening in vitro through in vivo and beyond, download, “Three Lessons to Help Accelerate Pharmaceutical Breakthroughs for CNS Disorders.”
About QPS Holdings, LLC
QPS is a global contract research organization (CRO) providing discovery, preclinical and clinical drug development services since 1995. The company accelerates pharmaceutical breakthroughs across the globe by delivering custom-built research services. An award-winning leader focused on bioanalytics and clinical trials, QPS is known for proven quality standards, technical expertise, a flexible approach to research, client satisfaction and turnkey laboratories and facilities. For more information, visit www.qps.com or email [email protected].
QPS Contact:
Media:
Kristin Jones
Tel.: +1 314-534-8187
Email: [email protected]




