The Birth Of RNAi
The Nobel Prize for the discovery that double-stranded RNAs (dsRNAs) can trigger silencing of complementary messenger RNA sequences was awarded in 1998, and the term ‘RNA interference’ (RNAi) was born. This discovery opened the door to a completely novel and untapped market within the pharmaceutical industry.
Shortly thereafter, short dsRNAs – or short interfering RNAs (siRNAs) – were generated artificially and used to demonstrate that this process also occurs in mammalian cells. The knowledge that small strands of RNAs can affect gene expression has had a tremendous impact on basic and applied research, and RNAi is currently one of the most promising new approaches for disease therapy. In 2004, only six years after the discovery of RNAi, the first siRNA-based human therapeutics – developed to treat wet age-related macular degeneration – entered Phase I clinical trials. RNAi is one of the fastest advancing fields in biology, and the flow of discovery gives true meaning to the expression ‘from the bench to the bedside’.
NEW IDEAS FOR RNAi THERAPEUTICS
Although there are still challenges, real strides towards realizing RNAi therapeutics for a wide range of diseases have been made. There are a host of reasons for optimism about the future of RNAi therapeutics and much has been accomplished in the race to bring this new treatment modality to the clinic. Ultimately, successful RNAi-based therapies for life-threatening or debilitating diseases will require the use of targeted delivery strategies that permit systemic delivery. Widespread interest in this phenomenon ensures that we will soon witness fast advances and new applications for RNAi-based therapies. Given the way RNAi has transformed basic research, and the speed with which it has reached the clinic, the coming years promise to be exciting. Ideas with world-changing potential might not be easy to realize, but people do it every day.
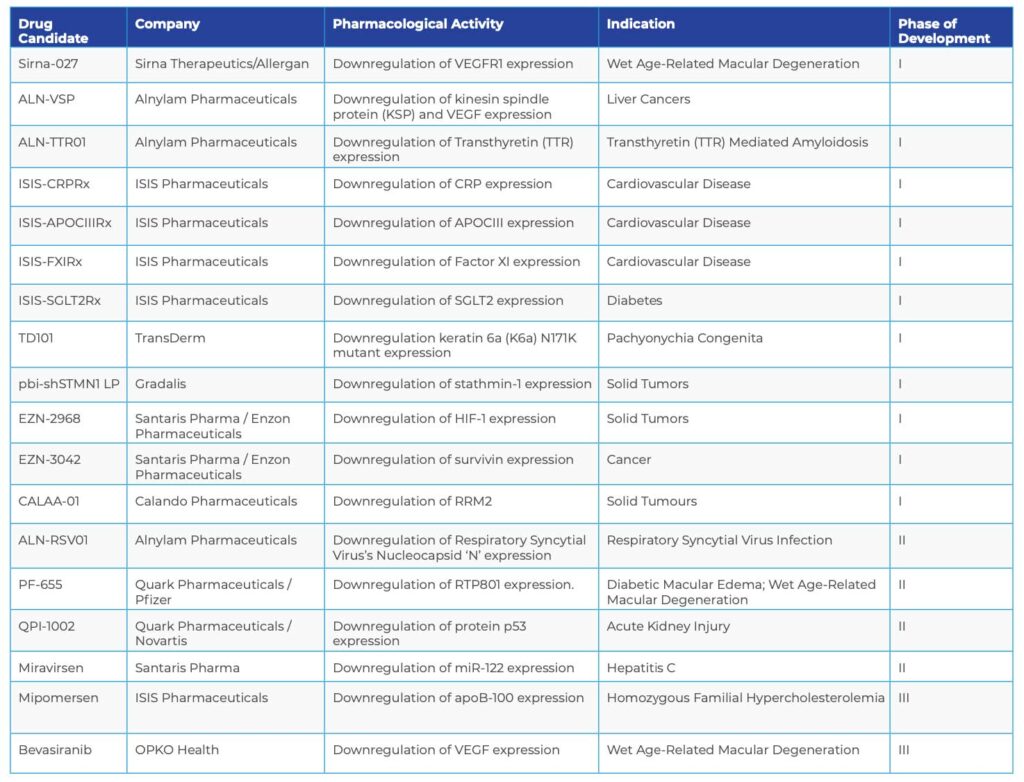
CURRENT siRNA-TARGETING STRATEGIES
For new technology, siRNAs have moved into the clinic at an unprecedented pace. Some examples of the diseases and siRNA-targeting strategies currently under investigation are described in the chart on the next page.
CHALLENGES
Although much is known about the mechanisms of RNAi, applications of this gene-silencing technology need to overcome a number of challenges. For one, RNAi is a fundamentally important regulatory mechanism in the cell, and tapping into it in the interests of therapeutic benefit could result in specific off-target effects. In addition, some synthetic siRNAs can induce general pro-inflammatory off-target effects.
Successful cell-specific delivery in vivo is clearly a crucial challenge for achieving the desired RNAi effect in clinical development. As with any other pharmacological treatment modality, therapeutic applications of siRNAs also require effective delivery to target cells and tissues. Most of the pending therapeutic applications based on RNAi rely on direct introduction of synthetic siRNAs to increase cellular uptake. The two main strategies are: delivery of chemically synthesized siRNAs (non-viral delivery), and delivery of siRNA-encoding genes by engineered viruses that will ultimately generate siRNAs by transcription within the target cells (viral delivery).
Until recently, most approaches to in vivo delivery have targeted local sites, such as the eye or lungs. Recent progress in the delivery of synthetic siRNAs to specific cell types in vivo strongly suggests that RNAi could potentially be used in the systemic treatment of a variety of diseases, including cancer, viral infections, autoimmune diseases, and neurodegenerative diseases.
QPS IS COMMITTED TO WORKING WITH YOU
QPS has extensive experience supporting RNAi therapeutic product development, and we truly understand its complexities, in particular the development of new imaging methods, such as quantitative whole-body autoradiography (QWBA) and microautoradiography (MARG) to monitor the in vivo biodistribution of siRNAs at the organ, tissue, and cellular level. In combination with reverse transcription PCR (RT-PCR), the up or down regulation of specific genes can be monitored at the tissue level. Furthermore, this can be coupled with PK sample analysis via hybridization-ELISA or LC-MS/ MS for PK/PD correlation. We are committed to working with you to advance your RNAi product portfolio in this rapidly developing market segment for the benefit of patients worldwide.
BROAD STRATEGIC ACCESS
QPS provides clients with broad strategic access to its nonclinical and clinical development capabilities, as well as its experience in conducting nonclinical and clinical development of a diverse portfolio of siRNAs across various therapeutic areas. Our preferred vendor agreements also provide for the establishment of a client-dedicated unit within our organization.
TIMELY DELIVERY
Partnering with QPS will position you to succeed in the timely delivery of your RNAi products to the marketplace.





