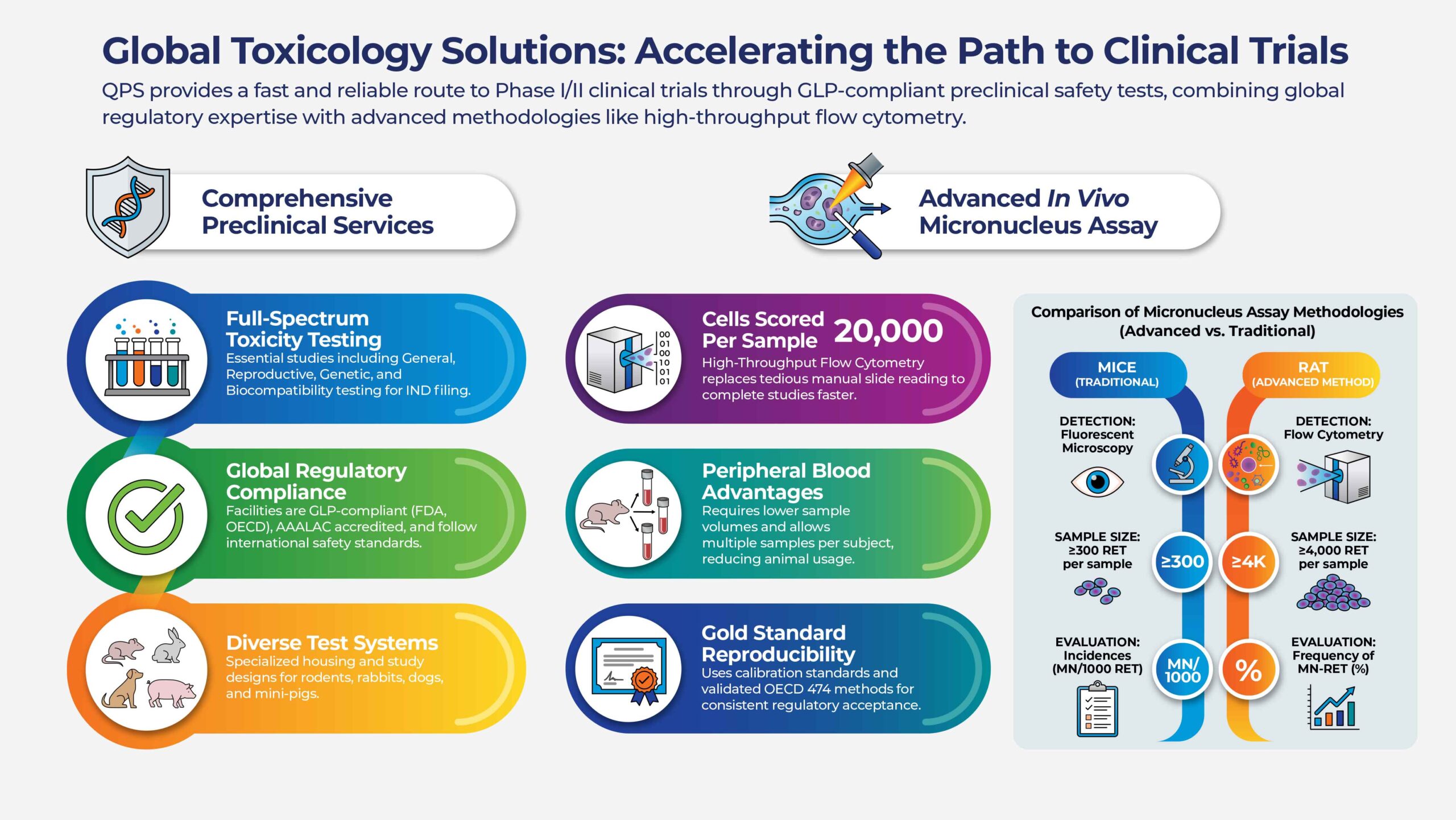
QPS Offers a wide range of toxicity studies

QPS’ globally recognized toxicology scientists offer a wide range of toxicity studies, as well as other preclinical safety tests, that are essential for preclinical drug development programs. We have a long and successful track record of designing specialized in vivo studies.
Quality and flexibility are the key factors as we work together to ensure that all regulatory studies for your investigational new drug (IND) filing are not overlooked. We will design and execute the most cost-effective development strategies for your drug candidates. Our commitment is to provide you with a fast and reliable route to clinical phase 1-2 studies.
Innovation in Genetic Toxicology
Revolutionize Your Safety Assessment with Advanced Micronucleus Assays
QPS is leading the way in Genetic Toxicity by utilizing state-of-the-art flow cytometry for In Vivo Micronucleus Assays (OECD 474). Genetic toxicology studies are conducted early in the safety testing program of your pharmaceutical and biotechnology products. QPS conducts genetic toxicology studies in a tiered approach, starting with an Ames test and progressing to the conduct of mammalian cell and in vivo assays, which are designed to assess the toxicological relevance of any earlier observations. All of our genetic toxicology studies are conducted in compliance to ICH guideline S2(R1).
Capabilities include:
- Gene Mutation Assays
- Ames Test
- In vitro Mouse Lymphoma Assay
- Cytogenetic Assays
- Chromosome Aberration Assay in Chinese Hamster Ovary (CHO) Cells
- In vitro Micronucleus Assays in CHO Cells
- In vivo Micronucleus Assay in Mouse and Rat
General Toxicology
- Single-Dose Toxicity Studies
- Repeated-Dose Toxicity Studies (subacute, subchronic, chronic studies)
- Systemic Safety and Risk Assessment
- Pharmacokinetic (PK) and Toxicokinetic (TK) studies
- Abnormal Toxicity Test
- Routes of administration: oral, intravenous (injection and infusion), intramuscular, subcutaneous, intraperitoneal, instillation, dermal, and others.
- Animal species: mice, rats, guinea pigs, hamsters, rabbits, dogs, and mini-pigs.
- Nonclinical study data package in compliance to CDISC Standard for Exchange of Nonclinical Data (SEND) for submission to FDA.
- Successfully completed over 2,200 GLP studies
- Successfully filed 58 USFDA IND applications
- Successfully filed 2 USFDA NDA applications
- Successfully filed 101 SEND packages to FDA with zero rejection
- Successfully completed 45 sponsor-initiated audits
Rodent peripheral blood or bone marrow erythrocytes are analyzed to detect micronuclei arising from structural or numerical chromosomal damage. Fluorescent staining and state-of-the-art flow cytometry enable automated identification and enumeration of micronucleated immature erythrocytes (reticulocytes) and mature erythrocytes. This approach provides a sensitive, quantitative assessment of in vivo genotoxic potential in accordance with OECD Test Guideline 474 (OECD TG 474).
Contact the QPS Business Development Today!
QPS Offers a Wide Range of Toxicity Studies

Rodent peripheral blood or bone marrow erythrocytes are analyzed to detect micronuclei arising from structural or numerical chromosomal damage. Fluorescent staining and state-of-the-art flow cytometry enable automated identification and enumeration of micronucleated immature erythrocytes (reticulocytes) and mature erythrocytes. This approach provides a sensitive, quantitative assessment of in vivo genotoxic potential in accordance with OECD Test Guideline 474 (OECD TG 474).
Benefits
- Detects Clastogenic and Aneugenic Effects:Identifies both chromosome breakage and chromosome loss events
- Quantitative & Objective Scoring:Automated analysis reduces subjectivity associated with manual microscopy
- High Statistical Power:Large cell counts per animal improve sensitivity and confidence
- Peripheral Blood Option:Supports repeated sampling and reduction in animal use
- Regulatory-Aligned Output:Generates data suitable for genetic toxicology risk assessment under OECD TG 474
- Regulatory Context:The in vivo mammalian erythrocyte micronucleus assay described in OECD TG 474 is a core component of genetic toxicology testing strategies and is accepted by global regulatory authorities for evaluating in vivo chromosomal damage.
Callouts
- Test Article Administration: Animals are exposed to the test article via an appropriate route and schedule to ensure systemic exposure, as required for in vivo genotoxicity assessment.
- Target Tissue Sampling: Peripheral blood or bone marrow is collected at defined sampling times to evaluate erythrocytes derived from exposed hematopoietic cells.
- Erythrocyte Differentiation: Fluorescent labeling distinguishes immature erythrocytes (polychromatic erythrocytes/reticulocytes) from mature erythrocytes.
- Micronucleus Detection: DNA-specific staining enables identification of micronuclei originating from acentric chromosome fragments or whole chromosomes not incorporated into the main nucleus.
- Flow Cytometric Enumeration: High-throughput flow cytometry scores large numbers of erythrocytes per animal, providing objective and statistically robust micronucleus frequency data.
- Data Interpretation: Results are evaluated for increases in micronucleated immature erythrocytes relative to controls, supporting assessment of clastogenic and aneugenic activity as described in OECD TG 474.
The QPS Advantage in Rat Micronucleus Testing:
- Higher Precision: While traditional mouse methods rely on manual microscopy of 300 cells, our rat peripheral blood assays use flow cytometry to routinely score 20,000 cells per sample.
- Speed & Efficiency: Our automated process allows for the analysis of an entire study in just one day, significantly reducing your project timelines.
- Animal Welfare & Integration: Using peripheral blood requires very low sample volumes and allows each subject to be sampled multiple times, which reduces the number of animals required and allows for easy integration into existing toxicology studies.
- Proven Reliability: Our methods include calibration standards to ensure reproducible data that is accepted by global regulatory agencies for GLP studies.
Reproductive Toxicology
QPS has been performing developmental and reproductive toxicity studies for pharmaceutical and biotechnology products for over 15 years. Following internationally recognized guidelines, including International Council of Harmonization (ICH), we offer overall developmental and reproductive toxicology program planning and individual study design for your pharmaceutical and biotechnology products.
Capabilities include:
- Fertility and Early Embryonic Development Studies
-
Teratology Studies (Embryo-fetal Development Studies)
Animal Species: Rat and Rabbit

Clinical Pathology
Our experienced Clinical Pathology Group offers a complete range of laboratory testing services. Our laboratories are equipped to provide the highest level of support to your toxicology programs through leading-edge technologies, quality assurance measurements, and laboratory management programs.
Capabilities include:
- Hematology (including coagulation)
- Serum Chemistry
- Urine Analysis

Histopathology
Microscopic evaluation of tissues, coupled with clinical pathology data, remains the cornerstone of toxicological evaluation of your pharmaceutical and biotechnology products. Our board-certified pathologists have decades of experience in the field with any type of study from animal model evaluation, through preliminary non-good laboratory practices (GLP) toxicity studies to formal GLP studies.
Capabilities include:
- Tissue and Slide Preparation
- Tissue Image
- Pathology Consultation and Evaluation
- Board Certified Histopathologist (DACVP)

Biocompatibility Test For Medical Devices
To support the biological evaluation of medical devices, we provide a range of tests following ISO-10993. With extensive experience in the study design and fast turn-around time, we provide high-quality data and reporting to meet your timeline for research and submission.
Capabilities include:
- Mutagenicity Test (ISO 10993-3)
- Cytotoxicity (ISO 10993-5)
- Implantation (ISO 10993-6)
- System Toxicity Test (ISO 10993-11)

Dermal Irritation And Contact Sensitization
Capabilities
- Irritation/Skin Sensitization Test (ISO 10993-10)
- Animal Species: Rodent and Rabbit
Local Tolerance Testing
- Evaluation of skin irritation and contact sensitizing of your test material is conducted by QPS to determine the potential to provoke ocular irritation, dermal irritation, or sensitization.
Wound Healing
In vivo animal models are the most efficient and clinically relevant approach for studying wound healing. Our veterinarians are capable of performing surgeries for testing of wound healing, with post-operation monitoring of clinical observations, body weight, and food consumption. Wounds are regularly measured for assessment of the extent of healing. At the completion of the in-life experimentation, our board-certified pathologist (DACVP) performs histopathology examination of the wound tissues. All studies are reviewed by IACUC in compliance of animal welfare requirements.
Capabilities include:
- In vivo wound healing test
- Animal Species: Rodent, Rabbit and Mini Pigs
Frequently Asked Questions
The in vivo mammalian erythrocyte micronucleus assay described in OECD Test Guideline 474 is a regulatory-accepted genetic toxicology test used to detect chromosomal damage in rodents. It measures micronuclei in immature and mature erythrocytes, indicating clastogenic (chromosome breakage) or aneugenic (chromosome loss) effects following systemic exposure to a test article.
Flow cytometry enables automated, high-throughput scoring of tens of thousands of erythrocytes per sample, significantly increasing statistical power compared to manual microscopy. This quantitative approach reduces subjectivity, improves reproducibility, and enhances sensitivity for detecting low-frequency micronucleus events in accordance with OECD TG 474.
The in vivo micronucleus assay detects both clastogenic effects (caused by chromosome breaks resulting in acentric fragments), and aneugenic effects (caused by whole chromosome loss during cell division). This dual detection capability makes it a core component of regulatory genetic toxicology testing strategies.
- Seamless integration into general toxicology studies
Flow cytometry-based assays routinely analyze ~20,000 erythrocytes per sample, compared to approximately 300 cells scored manually in traditional microscopy-based methods. This higher cell count improves data robustness, sensitivity, and confidence in genotoxicity risk assessment.
Data generated under OECD TG 474 are accepted by global regulatory agencies as part of standard genetic toxicology testing batteries for pharmaceuticals, chemicals, and other regulated products, including GLP-compliant safety assessments.
QPS combines state-of-the-art flow cytometry, calibrated reference standards, and experienced scientific leadership to deliver high-precision, regulatory-ready micronucleus data. Rat peripheral blood assays can be completed in as little as one day, offering faster timelines, reduced animal use, and reliable results suitable for global submissions.
Leadership
Testimonials

Contact Us
Ready to accelerate your drug development with QPS Toxicology services?
Contact us today to discuss how we can help you develop a custom-built research solution.
Visit: https://www.qps.com/
Set Up a Meeting: https://www.qps.com/contact
Email: info@qps.com



